Bristol Myers Squibb BMY lately introduced that it’s going to discontinue the late-stage Librexia research on cardiovascular candidate milvexian, an investigational oral, extremely selective issue XIa (FXIa) inhibitor.
BMY and associate Johnson & Johnson JNJ had been evaluating the efficacy and security of pipeline candidate milvexian when added to the usual of care (typical antiplatelet remedy) for sufferers after a latest acute coronary syndrome (ACS) occasion.
Nevertheless, BMY and JNJ determined to discontinue the section III Librexia ACS research following a preplanned interim evaluation by the Impartial Information Monitoring Committee (“IDMC”), which decided that the research is unlikely to satisfy the first efficacy endpoint.
The discontinuation of the section III Librexia ACS research was a setback, given the market potential.
The profitable growth of milvexian for this indication would have boosted BMY’s cardiovascular portfolio, which contains Camzyos (mavacamten), a first-in-class cardiac myosin inhibitor.
BMY obtained FDA approval of the drug in 2022 for the remedy of adults with symptomatic New York Coronary heart Affiliation class II-III obstructive hypertrophic cardiomyopathy (HCM) to enhance useful capability and signs.
Nonetheless, the IDMC suggested that the 2 different late-stage research — Librexia AF for atrial fibrillation (AF) and Librexia STROKE for secondary stroke prevention (SSP) — ought to proceed as deliberate. Prime-line knowledge from these research is anticipated in 2026.
BMY’s cardiovascular portfolio additionally contains blood thinner medication Eliquis, for which BMY has a worldwide co-development and co-commercialization settlement with pharma large Pfizer PFE. Eliquis is among the largest contributors to the highest line.
Competitors for BMY’s Cardiovascular Medicine
Cytokinetics CYTK is growing aficamten, an investigational, oral, small-molecule cardiac myosin inhibitor. CYTK is at the moment in search of FDA approval for aficamten for the remedy of sufferers with obstructive HCM in america.
In Could 2025, Cytokinetics introduced that the FDA has prolonged the goal motion date for the brand new drug software NDA in search of approval of aficamten for obstructive HCM to Dec. 26, 2025.
A possible approval of aficamten will pose competitors to Camzyos.
JNJ’s Xarelto can be a Issue Xa inhibitor, much like Eliquis.
Nevertheless, JNJ is dealing with patent challenges for Xarelto in america. JNJ co-developed Xarelto with Bayer AG.
BMY’s Value Efficiency, Valuation and Estimates
Shares of Bristol Myers have misplaced 19.1% 12 months so far in opposition to the trade’s development of 16.5%.
Picture Supply: Zacks Funding Analysis
From a valuation standpoint, BMY is buying and selling at a reduction to the large-cap pharma trade. Going by the worth/earnings ratio, BMY’s shares at the moment commerce at 7.55x ahead earnings, decrease than its imply of 8.41x and the large-cap pharma trade’s 16.84X.
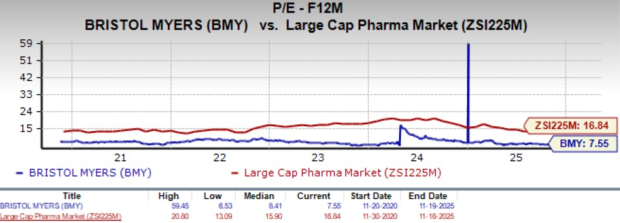
Picture Supply: Zacks Funding Analysis
The Zacks Consensus Estimate for 2025 earnings per share has moved north up to now 60 days, whereas that for 2026 EPS has moved south.
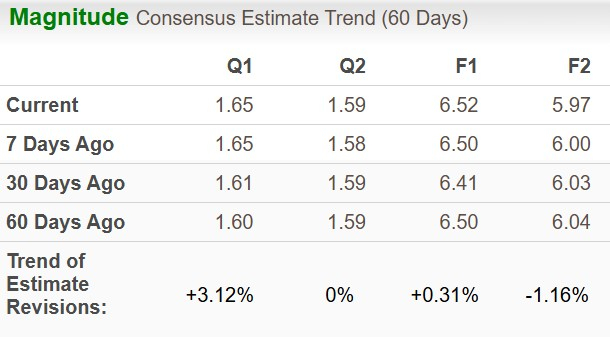
Picture Supply: Zacks Funding Analysis
BMY at the moment carries a Zacks Rank #3 (Maintain). You may see the full checklist of at the moment’s Zacks #1 Rank (Sturdy Purchase) shares right here.
Zacks’ Analysis Chief Names “Inventory Most More likely to Double”
Our staff of specialists has simply launched the 5 shares with the best likelihood of gaining +100% or extra within the coming months. Of these 5, Director of Analysis Sheraz Mian highlights the one inventory set to climb highest.
This prime decide is a little-known satellite-based communications agency. Area is projected to grow to be a trillion greenback trade, and this firm’s buyer base is rising quick. Analysts have forecasted a serious income breakout in 2025. In fact, all our elite picks aren’t winners however this one may far surpass earlier Zacks’ Shares Set to Double like Hims & Hers Well being, which shot up +209%.
Free: See Our Prime Inventory And 4 Runners Up
Bristol Myers Squibb Firm (BMY) : Free Inventory Evaluation Report
Johnson & Johnson (JNJ) : Free Inventory Evaluation Report
Pfizer Inc. (PFE) : Free Inventory Evaluation Report
Cytokinetics, Integrated (CYTK) : Free Inventory Evaluation Report
This text initially printed on Zacks Funding Analysis (zacks.com).
The views and opinions expressed herein are the views and opinions of the writer and don’t essentially replicate these of Nasdaq, Inc.







